The following 30-second video demonstrates how our technology works:
Intellectual Property
TheraBiome owns five issued US patents and 19 international patents on its proprietary platform technology. Patents protect drug and biologic delivery, including vaccines.
Differentiated Drug Delivery Target Product Profile (TPP)
• TheraBiome’s GEMICEL® platform technology is Phase II-ready and based on its strong science and human proof-of-concept clinical studies compliant with FDA and international regulatory standards.
• The platform is suitable for targeted oral delivery of small molecules and biologics, including vaccines to the colon or to both small intestine and colon.
• Longer acting and locally delivered
• TheraBiome is ready to support CMC stratify and deliverables
• Disease and therapeutic area agnostic despite special focus on GI-related diseases.
• Easy to swallow – patient compliant products
Phase 2-Ready Precision GI Targeting
GEMICEL® Delivery System
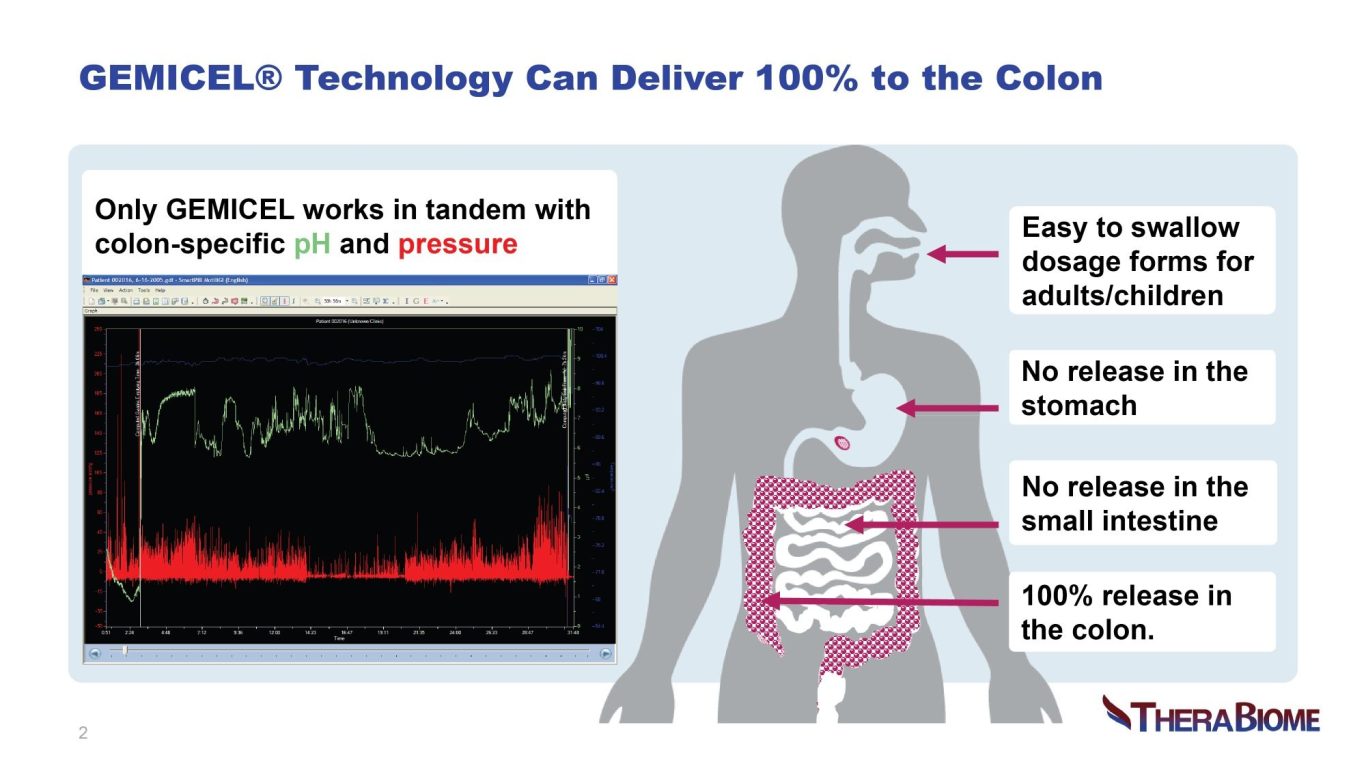
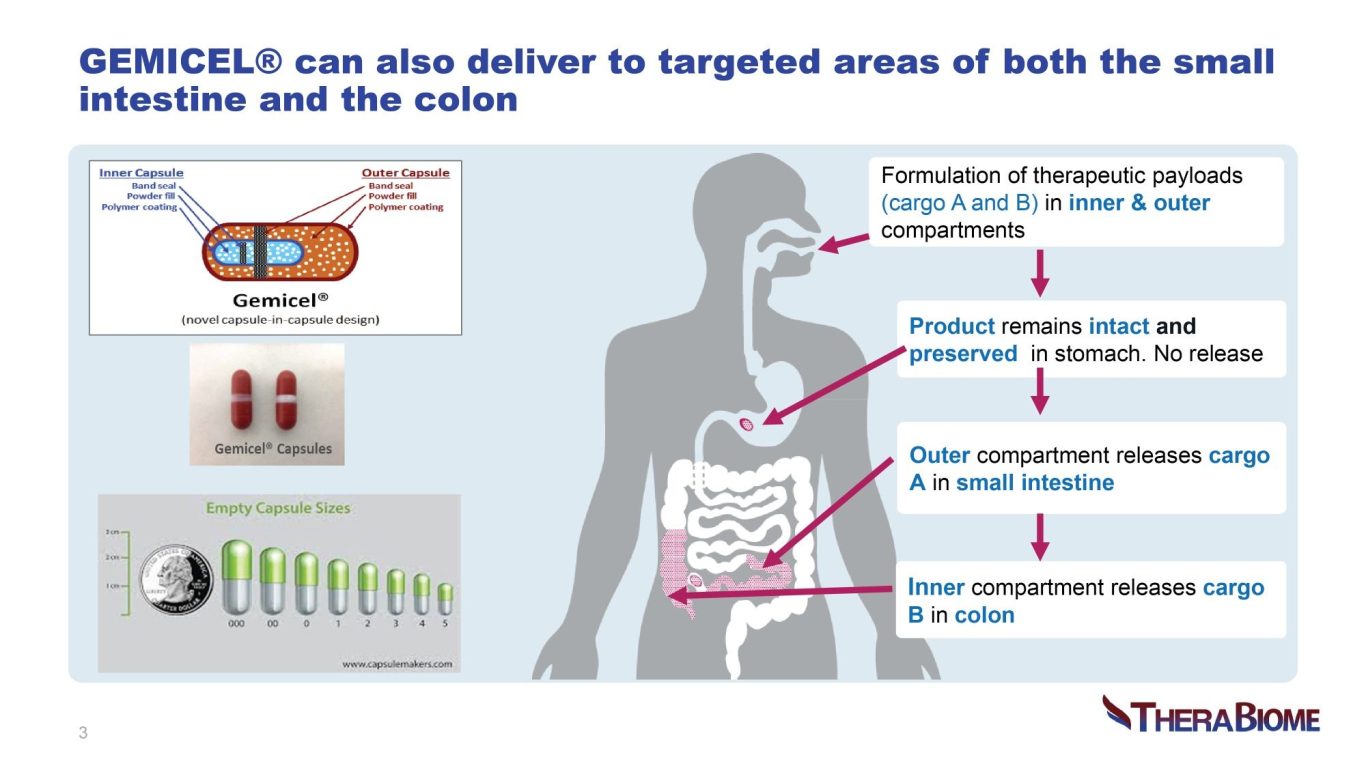
The gastrointestinal (GI) tract’s heterogeneous pH and chemical milieu poses challenges to ensuring targeted delivery of therapeutics into physiologically relevant gut segments using traditional technology. Some therapeutics may be ineffective when taken orally; they may be too sensitive, incompatible, subject to high first-pass metabolism or require higher doses. As a result, the formulation may fail to demonstrate efficacy or safety. GEMICEL enables precise and sitespecific targeted delivery with controlled release using a proprietary oral formulation, maximizing chances of success in meeting target product profile in clinical trials. All formulation components are safe, and the technology has been successfully scaled up. TheraBiome is seeking partners who may benefit from precision GI targeting.
Phase 2-Ready Precision GI Targeting (Cont’d)
Phase 2 Ready
Scintigraphy studies were conducted in 27 healthy volunteers. GRAS scale-up was completed. Phase I and IB studies were initiated in 24 subjects before Abbvie ended the ulcerative colitis study following AbbVie’s acquisition of Allergan.
Phase 2-Ready Precision GI Targeting (Cont’d)
Unmet Medical Need: Oral Targeted Enteral Delivery
Targeted oral delivery technologies include conventional pH-sensitive enteric coatings, multimatrix, osmotic-controlled and timed-release methods. Limitations include variable or off-target delivery, reduced potency, leakage from one site to another, lack of patent protection and manufacturing complexity, as well as high COGS. GEMICEL overcomes these challenges costeffectively based on its single- or dual-targeted drug delivery platform. Human clinical scintigraphy studies confirmed that GEMICEL is highly tunable and reliably delivers high therapeutic payloads to the colon alone, or to a combination of the small intestine and the colon. This patented delivery technology is based on dual polymer layers programmed to dissolve at specific pH and peristaltic pressure levels. GEMICEL in various oral forms delivers small molecules and biologics including proteins, peptides, live biotherapeutics and vaccines (providing mucosal immunity) to specific target locations in the GI tract. This technology is perfectly designed to target the microbiome with products which are otherwise struggle to achieve their intended effects. As a innovation it effectively addresses unmet medical needs across multiple-therapeutic areas.
